Explained: Why the Earth isn't an ice cube
(4 min read) The basic atmospheric science behind warming
Last week we covered why we all need to take action. This week, we’ll start with a basic understanding of the Greenhouse Gas Effect. Need a TL;DR? Watch this video.
July 1969. After 4 days in space, humans prepare to set foot on the moon for the first time. The climate, however, is unforgiving. Surface temperatures swing over 500F from -280F to 260F over the long 708 hour lunar day. The astronauts' only hope for survival is their specialized suits to reflect new heat away and trap existing heat inside (among other space hazards). In other words, to make the moon feel more like home.

But why is this the case? Why isn’t the moon more like the Earth? Both the Earth and the Moon get their energy entirely from the sun. Both are small rocks hurtling around the sun at roughly the same distance. What allows Earth to maintain a stable, comfortable climate suitable for squishy mammals like us?
Heating Up: How the sun gives us energy
First, let’s consider the source: the Sun. The sun is a massive collection of hydrogen atoms, squished together by gravity, that bond to create helium in a process known as nuclear fusion. This process releases an absurd amount of energy equivalent to ~2,000,000,000 nuclear bombs per second, otherwise known as a fuck-ton (FT).

This energy radiates outwards into space towards all objects (like the moon and Earth). We can measure the energy released by the Stefan Boltzman equation and classify the type of energy released on a Blackbody spectrum. <Eyes glaze over.. I thought you said ‘simple’.>
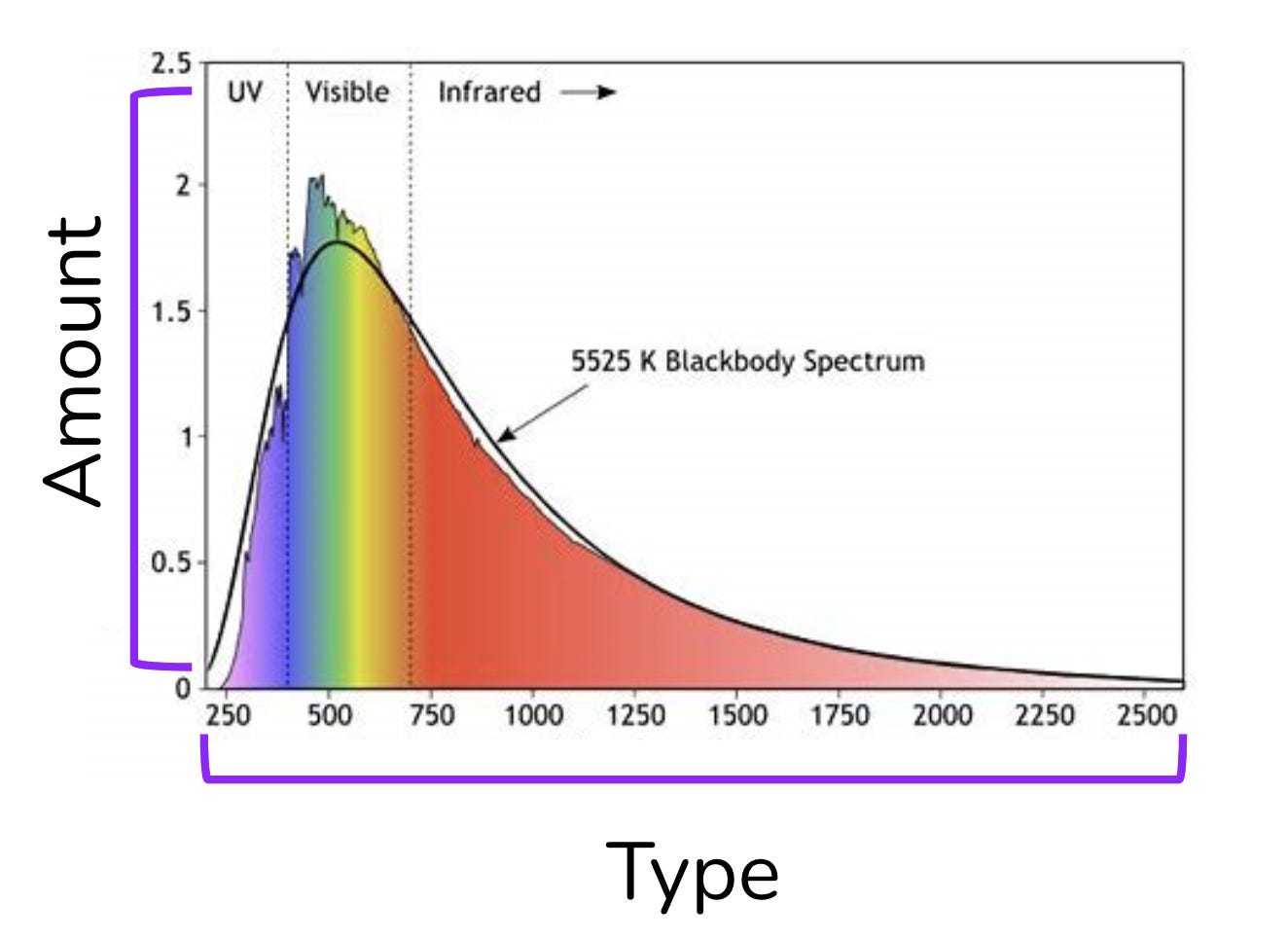
Without drowning in the math, we can precisely predict the amount and the type of energy the sun will bestow upon an object at any distance. Over a period of time, the amount of energy absorbed will reach equilibrium with the amount of energy released. ‘Equilibrium energy’ is a fancy way of saying ‘average temperature’. Put simply, we can calculate the temperature of an object based on its distance from the sun.
A Normal Rock: Why the moon is so extreme
A rather normal rock, it orbits the earth as the earth orbits the sun. Like all objects, the moon heats up as it absorbs heat (radiation) from the exploding sun. After it heats up, it radiates energy directly back out into space around it. Crunching the numbers, we net out at an average temperature of ~0F which is precisely the observed average temperature of the moon.
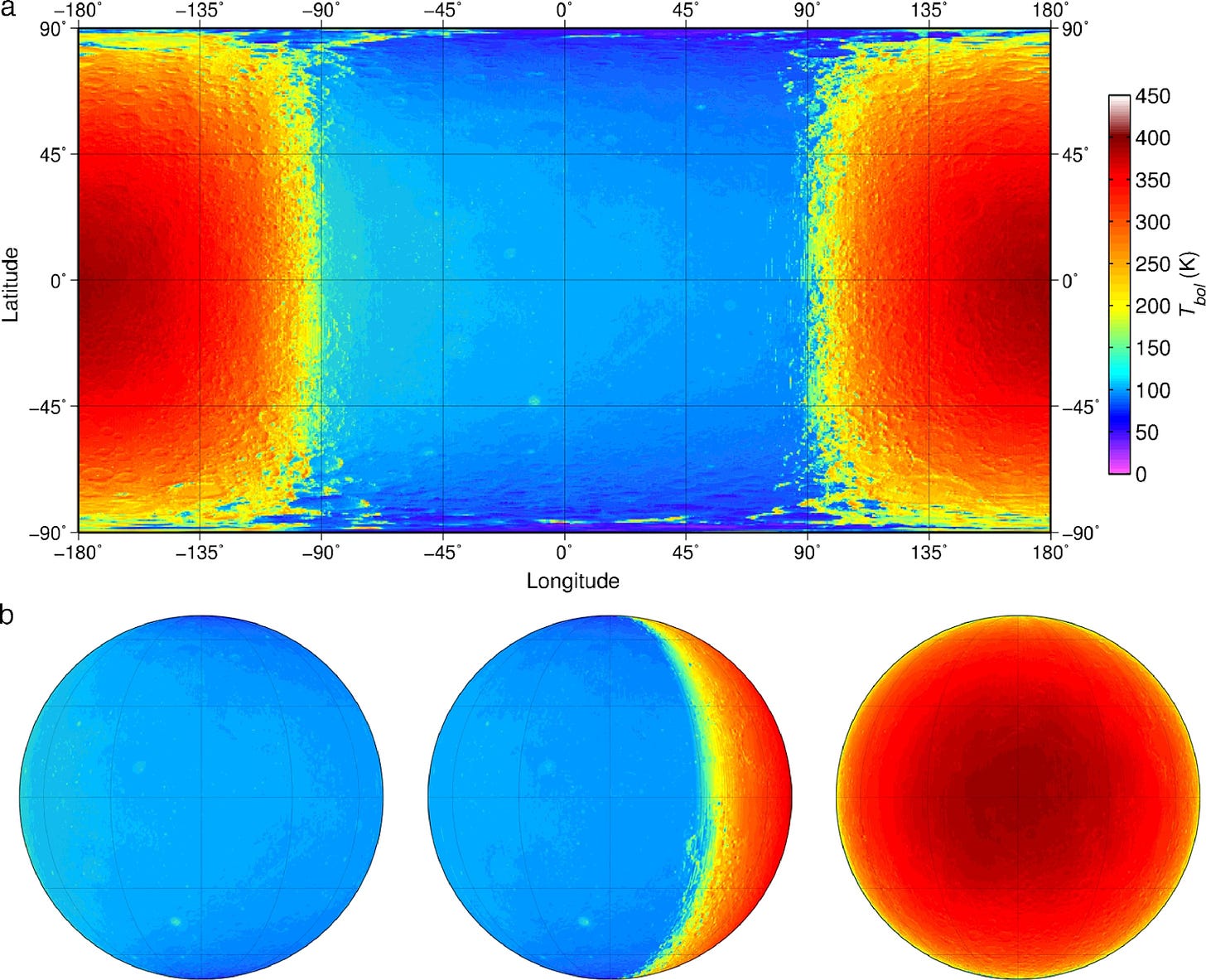
So a normal rock at about our distance from the sun is searing hot and apocalyptically cold. So why doesn’t the earth experience sharp spikes of +/- ~250F every day? While the moon is just a rock1, the earth is surrounded by a blanket of gases called an atmosphere. These gases act as a bouncer of sorts, deciding which energy comes in and out.
A Fancy Rock: Earth and her cozy sweater
Unlike the moon, our atmospheric particles reflect some types of sun energy immediately back out into space, while letting other types pass unnoticed to the surface.
Revisiting our trusty blackbody spectrum, most energy emitted from the sun reaches Earth as visible light, which passes right through the atmosphere, enabling plants to grow & make oxygen for us to breathe. How generous.
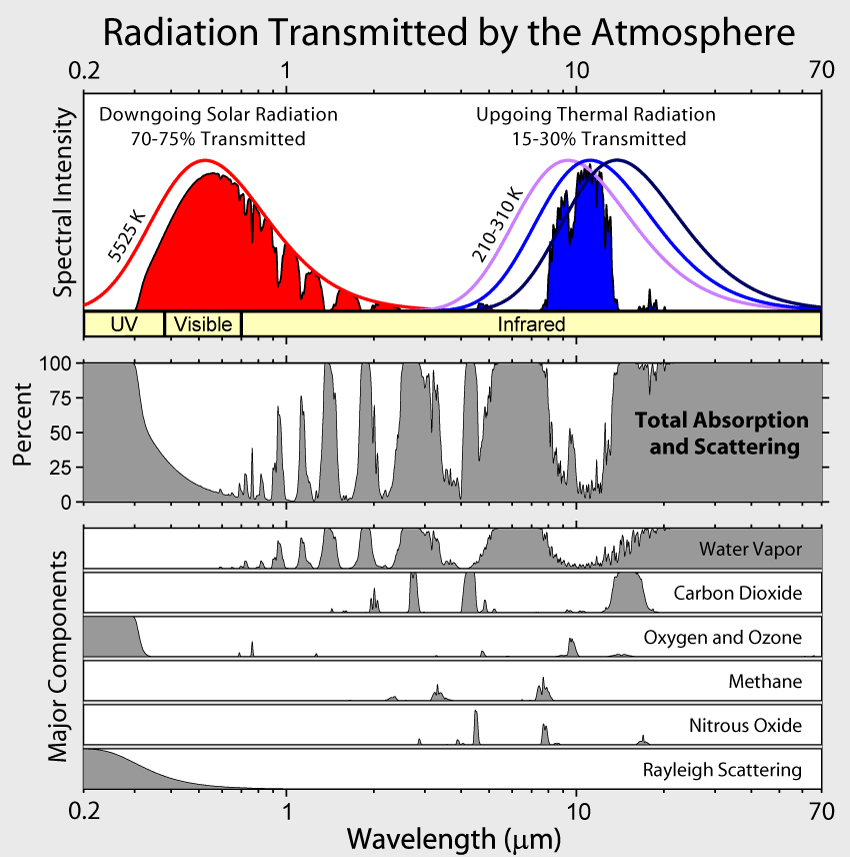
However, after heating the surface, this energy is then emitted back towards space as infrared (like hot pavement on a summer day). The atmosphere contains particles (like water vapor and CO2) that are awesome at absorbing that infrared energy and re-reflect some energy back towards earth!
The net effect is less extreme highs (from blocking some of the inbound) and less extreme lows (from trapping the rebound).
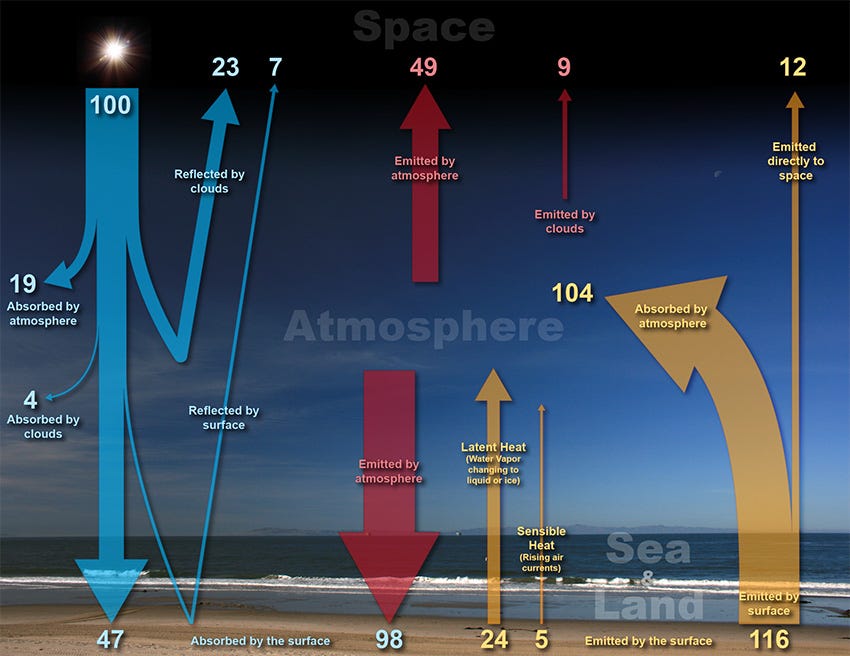
If it’s reflected back towards Earth, the average surface temperature of Earth rises. This is known as the greenhouse effect which brings the Earth’s temperature into equilibrium.
A Delicate Dance… until now
For nearly the last million years, these particles have maintained a stable equilibrium. Letting just enough energy in and just enough out enabling life (like ours) to adapt to this comfortable habitable zone.
While these molecules are essential for regulating our temperature, we’ve recently dumped a lot more of them into the atmosphere very quickly. Greenhouse gas concentrations are at the highest point for the last 800,000 years.
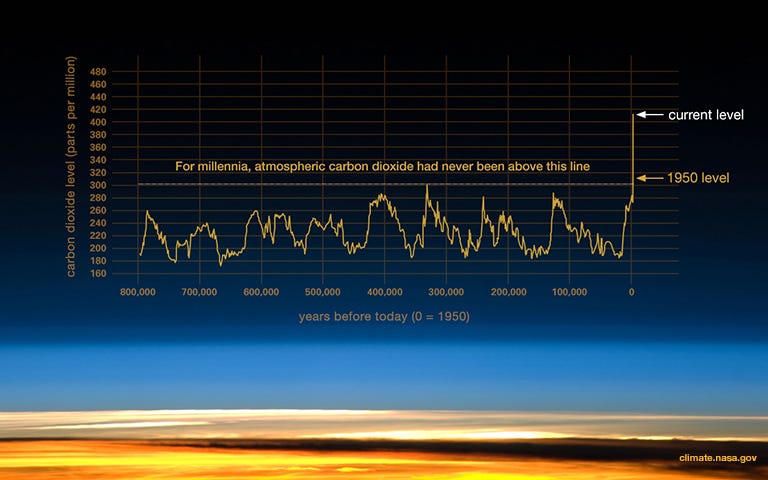
More greenhouse gas particles = more energy trapped = more embedded heat = higher average temperatures.
Humans have never done this before; we’re in (literally) uncharted territory. It’s the greatest experiment our species has ever attempted, but what happens if it goes awry?
Unfortunately, it already has…
Up next: What happens as we crank up the thermostat on Mother Earth?
But first… How are we so sure it’s human-emitted GHG and not natural causes?
Enjoy this article? Consider subscribing!
Already subscribed? Share it with your friends, family, coworkers.
Until next time,
Obsessed and want to learn more? Here are my favorite sources for this article:
Like it: Awesome 3 minute explainer
Love it: More technical 20 minutes explainer
Gotta have it: 39 hour University of Chicago Course from Coursera (source of truth for me)
Usual disclaimers: I’m not an expert and will never claim to be. I’ll probably be lacking context, too vague, or flat-out wrong frequently & I hope folks will hold me accountable. After all, the fastest way to find the right answer is to post the wrong answer visibly on the Internet. Lastly, there is a ton of great information online already (e.g. Drawdown, Breakthrough Energy playbooks). I will synthesize and cite as I go.
it has a very thin atmosphere





